
Total citations >6400; 11 papers > 100 citations.
Preprints of submitted articles
Research Papers
Gold(I) Dimers with Hemilabile Azophosphine Ligands in Catalysis, E. J. Jordan, B. L. Greene, L. E. English, Ellen M. Tait, P. W. Davies* and A. R. Jupp* Dalton Trans. 2026 ASAP
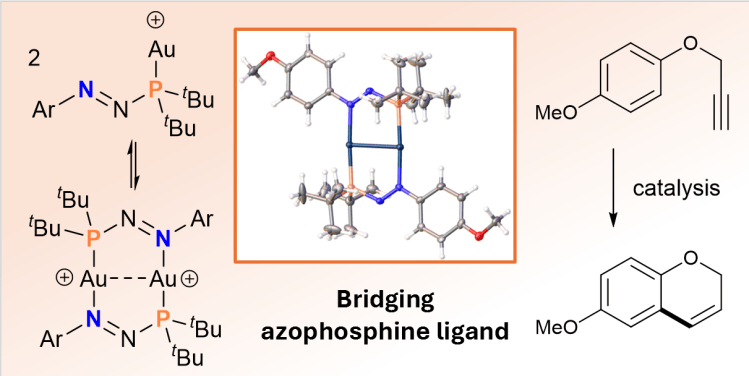
Tuning the Electronic Properties of Azophosphines as Ligands and their Application in Base-Free Transfer Hydrogenation Catalysis E. J. Jordan, E. D. E. Calder, B. L. Greene, H. V. Adcock, L. Male, P. W. Davies and A. R. Jupp* Organometallics 2024, 43, 2674–2685.
ChemRxiv version 08 July, 2024
Regiodivergent gold-catalyzed rearrangement-addition reactions of sulfenylated propargylic carboxylates with indoles N. Y. More, P. A. Rist, A. Gupta, P. W. Davies* Org. Lett. 2024, 26, 7713-7717. OPEN DATA.

Invited contribution to Special Issue “Gold Mediated Chemistry”
Regiodivergent Synthesis of 4- and 5-Sulfenyl Oxazoles from Alkynyl Thioethers P. Sekar, A. Gupta, L. E. English, C. E. Rabbitt, L. Male, A. R. Jupp* and P. W. Davies* Chem. Eur. J. 2024, 30, e202401465. OPEN DATA.

Designated as a Very Important Paper
Highlighted in “Hot Topic: Gold“
A laterally-fused N-heterocyclic carbene framework from polysubstituted amino-imidazo[5,1-b]oxazol-6-ium salts A. D. Gillie, M. G. Wakeling, B. L. Greene, L. Male and P. W. Davies* Beilstein J. Org. Chem. 2024, 20, 621-627. OPEN DATA.
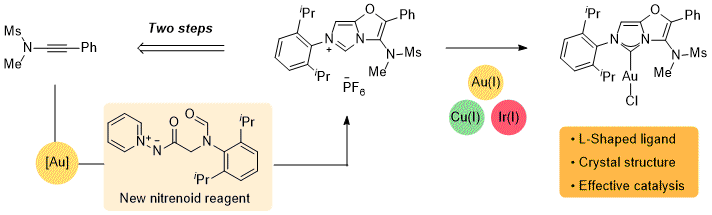
Invited submission for ISySyCat special issue
Synthesis of α-tertiary primary amines and 1,2-amino alcohols from vinyl azides by light induced denitrogenative alkylarylation/dialkylation S. Li, H.-W. Du, P. W. Davies* and W. Shu* CCS Chemistry 2023, 6, 1060-1070.
ChemRxiv version 24 Apr, 2023

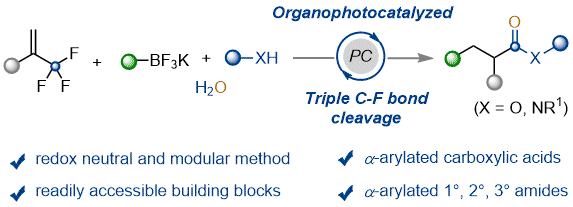
Modular Synthesis of α-Arylated Carboxylic Acids, Esters and Amides via Photocatalyzed Triple C−F Bond Cleavage of Methyltrifluorides, S. Li, P. W. Davies* and W. Shu* Chem. Sci. 2022, 13, 6636-6641.
Gold-Catalyzed Annulations with Nucleophilic Nitrenoids Enabled by Heteroatom-Substituted Alkynes, P. W. Davies* Chem. Rec. 2021, 21, 3964-3977.
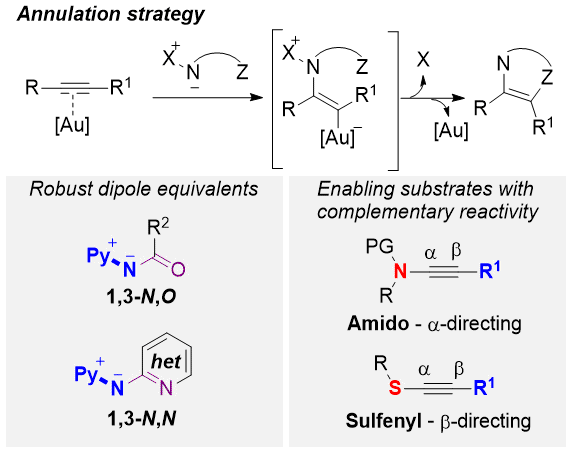
Invited submission to as special issue Recent Advances in Transition‐Metal Catalysis
A Radical-Initiated Fragmentary Rearrangement Cascade of Ene-Ynamides to [1,2]-Annulated Indoles via Site-Selective Cyclization, S. Li, Y. Wang, Z. Wu, W. Shi, Y. Lei, P. W. Davies* and W. Shu* Org. Lett. 2021, 23, 7209-7213.
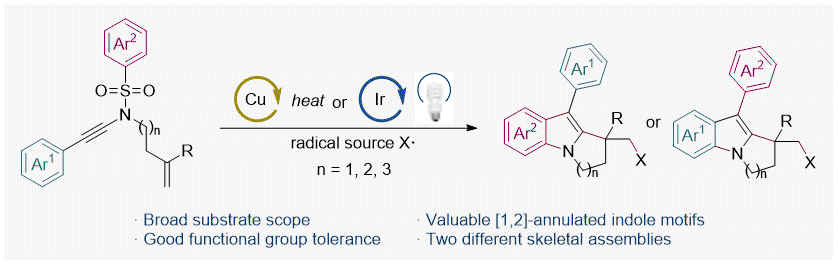
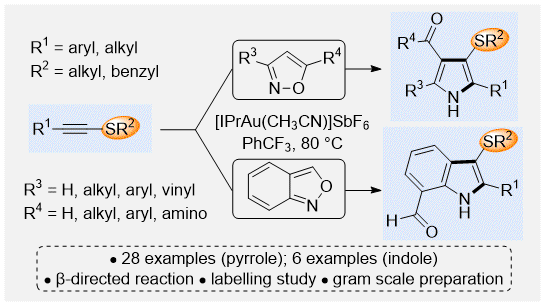
Gold(I)-Catalyzed Synthesis of 3-Sulfenyl Pyrroles and Indoles by a Regioselective Annulation of Alkynyl Thioethers, P. E. Simm, P. Sekar, J. Richardson and P. W. Davies* ACS Catalysis 2021, 11, 6357–6362. OPEN DATA.
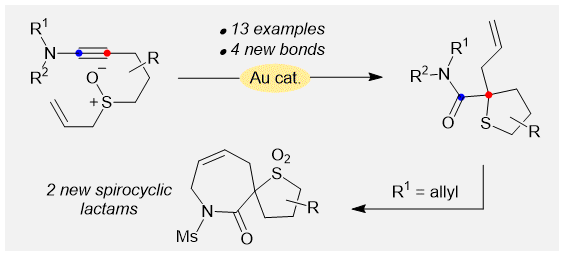
Gold-catalysed cycloisomerisation of ynamides to access 2,2-disubstituted tetrahydrothiophene motifs, P. Heer Kaur and P. W. Davies* Synlett 2021, 32, 897-900. Link
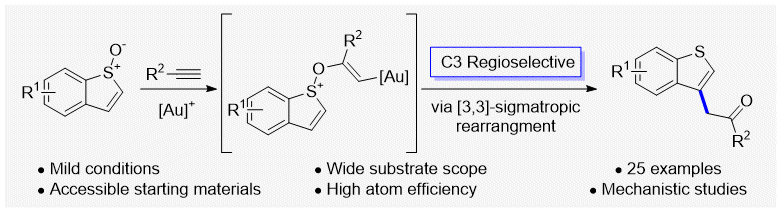
Gold-Catalyzed Intermolecular Alkyne Oxyarylation for C3-Functionalization of Benzothiophenes, P. A. Rist, R. S. Grainger* and P. W. Davies*, Org. Lett. 2021, 23, 642–646. OPEN DATA.
Diastereoselective sulfur ylide rearrangements from gold catalysed oxidation of
ynamides, J. D. Priest, L. Male and P. W. Davies* Tetrahedron 2021, 78, 131757. Free access
Special issue in memory of Professor Jonathan Williams
Sulfenyl Ynamides in Gold Catalysis: Synthesis of Oxo-functionalised 4-Aminoimidazolyl Fused Compounds by Intermolecular Annulation Reactions, E. M. Arce, S. G. Lamont, P. W. Davies,* Adv. Synth. Catal. 2020, 362, 2503-2509.
Balancing Bulkiness in Gold(I) Phosphino-triazole Catalysis, Y. Zhao, M. G. Wakeling, F. Meloni, T. J. Sum, H. van Nguyen, B. R. Buckley, P. W. Davies, J. S. Fossey,* Eur. J. Org. Chem. 2019, 31-32, 5540-5538.
Invited submission to special issue: Heterocyclic Chemistry Eds. T. Ooi, O. Reiser, A. Smith, C. Vanderwal
Synthesis of Thiomorpholin‐3‐ones by a Gold‐Catalysed Oxidative Cyclisation‐Rearrangement Cascade from Ynamides, T. E. Baker, P. W. Davies,* Eur. J. Org. Chem. 2019, 31-32, 5201-5204.
Invited submission to special issue: Heterocyclic Chemistry Eds. T. Ooi, O. Reiser, A. Smith, C. Vanderwal
A Gold Carbene Manifold to Prepare Fused γ‐Lactams by Oxidative Cyclisation of Ynamides, F. Sanchez-Cantalejo, J. D. Priest, P. W. Davies,* Chem. Eur. J. 2018, 24, 17215-17219.
Synthesis of N-Acyl Pyridinium-N-Aminides and Their Conversion to 4-Aminooxazoles via a Gold-Catalyzed Formal (3+2)-Dipolar Cycloaddition, M. P. Ball-Jones, P. W. Davies,* Org. Synth. 2018, 95, 112-126.
Alkynyl thioethers in gold-catalysed annulations to form oxazoles, R. J. Reddy, M. P. Ball-Jones, P. W. Davies,* Angew. Chem. Int. Ed. 2017, 56, 13310-13313.
Alkynyl sulfoxides as α-sulfinyl carbene equivalents: Gold-catalysed oxidative cyclopropanation, M. J. Barrett, G. F. Khan, P. W. Davies,* R. S. Grainger* Chem. Commun. 2017, 53, 5733-5736.
General Entry into o-,o’-Heteroatom-Linked N-(Hetero)aryl Imidazole Motifs by Gold-Catalysed Formal [3+2]-Dipolar Cycloaddition, M. Garzón, E. M. Arce, R. J. Reddy, P. W. Davies,* Adv. Synth. Catal. 2017, 359, 1837-1843.
Designated as a Very Important Publication
In the top 25 Most Accessed ASC papers in 2017
Efficient and flexible synthesis of highly functionalised 4-amino oxazoles by a gold-catalysed intermolecular formal [3+2]-dipolar cycloaddition, A. D. Gillie, R. Jannapu Reddy, P. W. Davies,* Adv. Synth. Catal. 2016, 358, 226-239.
Designated as a Very Important Publication
#6/25 Most accessed ASC paper in 2016 and still in the top 25 Most accessed ASC papers in 2018/9.
Divergent C-H Insertion-Cyclization Cascades of N-Allyl Ynamides; H. V. Adcock, E. Chatzopoulou, P. W. Davies* Angew. Chem. Int. Ed. 2015, 54, 15525-15529.
Regioselective functionalisation of dibenzothiophenes through gold-catalysed intermolecular alkyne oxyarylation, M. J. Barrett, P. W. Davies,* R. S. Grainger,* Org. Biomol. Chem. 2015, 13, 8676-8686.
A Direct Route into Fused Imidazo-diazines and Imidazo-pyridines Using Nucleophilic Nitrenoids in a Gold-Catalyzed Formal [3 + 2]-Dipolar Cycloaddition; M. Garzon, P. W. Davies,* Org. Lett. 2014, 16, 4850-4853.
Highlighted in Synfacts 2014, 14, 1246.
Highlighted in Organic Chemistry Portal (https://www.organic-chemistry.org/abstracts/lit4/661.shtm)
1,2-N-Migration in a Gold-Catalysed Synthesis of Functionalised Indenes by the 1,1-Carboalkoxylation of Ynamides; H. V. Adcock, T. Langer, P. W. Davies,* Chem. Eur. J. 2014, 20, 7262-7266.
A Gold-Catalysed Fully Intermolecular Oxidation and Sulfur-Ylide Formation Sequence on Ynamides; M. Dos Santos, P. W. Davies,* Chem. Commun. 2014, 50, 6001-6004.
Highly regioselective synthesis of 2,4,5-(hetero)aryl substituted oxazoles by intermolecular [3+2]-cycloaddition of unsymmetrical internal alkynes; E. Chatzopoulou, P. W. Davies,* Chem. Commun. 2013, 49, 8617-8619.
A reactivity switch in the gold-catalysed coupling of allyl sulfides with propargylic carboxylates; P. W. Davies,* S. J. C. Albrecht, Synlett, 2012, 23, 70-73.
Intermolecular and selective synthesis of 2,4,5-trisubstituted oxazoles by a gold-catalysed formal [3+2] cycloaddition; P. W. Davies,* A. Cremonesi, L. Dumitrescu, Angew. Chem. Int. Ed. 2011, 38, 8931-8935.
Selected as a Hot Article by the editors of Angewandte Chemie for its importance in a rapidly evolving field of high current interest.
Highlighted in Synfacts 2011, 11, 1174.
Isotopic labelling studies for a gold-catalysed skeletal rearrangement of alkynyl aziridines; P. W. Davies,* N. Martin, N. Spencer, Beilstein J. Org. Chem. 2011, 7, 839-846.
Invited contribution to special issue: Gold Catalysis. Ed. F. D. Toste.
An efficient and selective synthesis of 2,5-substituted pyrroles by gold-catalysed ring expansion of alkynyl aziridines; P. W. Davies,* N. Martin, J. Organomet. Chem. 2011, 696, 159-164.
Invited contribution to special issue: Catalytic addition of E-H bonds to non-activated carbon-carbon multiple bonds. Ed. G. Bertrand.
Site-specific introduction of gold-carbenoids by intermolecular oxidation of ynamides or ynol ethers; P. W. Davies,* A. Cremonesi, N. Martin, Chem. Commun. 2011, 47, 379-381.
Invited contribution to “Emerging Investigators 2011” issue
Gold-catalysed room-temperature cycloisomerisation of alkynes and unactivated enolisable ketones; P. W. Davies,* C. Detty-Mambo, Org. Biomol. Chem. 2010, 8, 2918-2922.
Alkynes as Masked Ylides Under Noble Metal Catalysis; P. W. Davies,* Pure Appl. Chem. 2010, 82, 1537-1544.
Gold or Platinum-Catalysed Synthesis of Sulfur Heterocycles: Access to Sulfur Ylides Without Using Sacrificial Functionality; P. W. Davies,* S. J. C. Albrecht, Angew. Chem. Int. Ed. 2009, 48, 8372-8375.
Counterion Effects in a Gold-Catalysed Synthesis of Pyrroles from Alkynyl Aziridines; P. W. Davies,* N. Martin, Org. Lett. 2009, 11, 2293-2296.
Highlighted in Organic Chemistry Portal
https://www.organic-chemistry.org/abstracts/lit2/556.shtm
Silver-Catalysed Doyle-Kirmse Reaction of Allyl and Propargyl Sulfides; P. W. Davies,* S. J. C. Albrecht, G. Assanelli, Org. Biomol. Chem. 2009, 7, 1276-1279.
Alkynes as Masked Ylides: Gold-Catalysed Intermolecular Reactions of Propargylic Carboxylates with Sulfides; P. W. Davies,* S. J. C. Albrecht, Chem. Commun. 2008, 238-240.
Total Synthesis of the Antibiotic Erypoegin H and Cognates by a PtCl2-Catalyzed Cycloisomerization Reaction; A. Fürstner,* E. K. Heilmann, P. W. Davies, Angew. Chem. 2007, 119, 4844-4847; Angew. Chem. Int. Ed. 2007, 46, 4760-4763.
Heterocycles by PtCl2-Catalyzed Intramolecular Carboalkoxylation or Carboamination of Alkynes; A. Fürstner,* P. W. Davies, J. Am. Chem. Soc. 2005, 127, 15024-15025.
Bidentate Ruthenium Vinylcarbene Catalysts Derived from Enyne Metathesis; A. Fürstner,* P. W. Davies, C. W. Lehmann, Organometallics, 2005, 24, 4065-4071.
Cyclobutenes by Platinum-Catalyzed Cycloisomerization Reactions of Enynes; A. Fürstner,* P. W. Davies, T. Gress, J. Am. Chem. Soc. 2005, 127, 8244-8245.
Asymmetric synthesis of avenaciolide via cascade palladium catalysed cyclisation–carbonylation of bromodienes; V. K. Aggarwal,* P. W. Davies, A. T. Schmidt, Chem. Commun. 2004, 1232-1233.
Palladium catalysed cyclisation–carbonylation of enynes to give cyclic γ,δ-unsaturated acids; V. K. Aggarwal,* M. Butters, P. W. Davies, Chem. Commun. 2003, 1046-1047.
A palladium catalysed cyclisation–carbonylation of bromodienes: control in carbonylation over facile β-hydride elimination; V. K. Aggarwal,* P. W. Davies, W. O. Moss, Chem. Commun. 2002, 972-973.
Reviews
Gold-Catalyzed Annulations with Nucleophilic Nitrenoids Enabled by Heteroatom-Substituted Alkynes, P. W. Davies* Chem. Rec. 2021, 21, 3964-3977.
1-(2-Pyrimidinylamino)pyridinium Inner Salt, P. W. Davies* e-EROS Encyclopedia of Reagents for Organic Synthesis, 2017.
Nucleophilic Nitrenoids Through π-Acid Catalysis: Providing a Common Basis for Rapid Access into Diverse Nitrogen Heterocycles; P. W. Davies,* M. Garzon, Asian J. Org. Chem. 2015, 4, 694-708.
π-Acid Mediated Insertion of Alkynes into Carbon-Heteroatom σ-Bonds; H. V. Adcock, P. W. Davies,* Synthesis, 2012, 44, 3401-3420.
Bürgenstock 2010: Stereochemistry on the shores; P. W. Davies,* Nature Chemistry, 2010, 2, 1011-1013.
Catalytic Carbophilic Activation: Catalysis by Platinum and Gold π Acids; A. Fürstner,* P. W. Davies,* Angew. Chem. 2007, 119, 3478-3519; Angew. Chem. Int. Ed. 2007, 46, 3410-3449.
Alkyne Metathesis; A. Fürstner,* P. W. Davies, Chem. Commun. 2005, 2307-2320.
Book Chapters
The Chemistry of Ynamides: Development, Syntheses, and Applications in Organic Synthesis; Chapter 7: Ynamides as precursors of carbenes and carbenoids; J. D. Priest, P. W. Davies; G. Evano and J. Zhao (Eds), Jenny-Stanford, Singapore, 2024, 369–468.
Metathesis in Natural Product Synthesis: Strategies, Substrates and Catalysts; Chapter 7: Ring-closing Alkyne Metathesis in Natural Product Synthesis; P. W. Davies; C. Meyer, S. Arseniyadis, J. Cossy (Eds), Wiley-VCH, Weinheim, 2010, 205-224.
Organometallics: Transition Metals in Organic Synthesis; P. W. Davies,* Annu. Rep. Prog. Chem. Sect. B: Org. Chem. 2010, 106, 98-119.
Handbook of Cyclization Reactions; Chapter 12: Ring-closing Metathesis of Alkynes; P. W. Davies; S. Ma (Ed), (Vol 1) Wiley-VCH, Weinheim, 2009, 599-620.
Organometallics: Transition Metals in Organic Synthesis; P. W. Davies,* Annu. Rep. Prog. Chem. Sect. B: Org. Chem. 2009, 105, 93-112.
Organometallics: Transition Metals in Organic Synthesis; P. W. Davies,* Annu. Rep. Prog. Chem. Sect. B: Org. Chem. 2008, 104, 68-87.
Misc.
For an interview with Paul see https://www.thieme-connect.com/ejournals/pdf/10.1055/s-0031-1291061.pdf
Iridium Catalysed Synthesis of Vinyl Ether by the Reaction of Alcohol and Vinyl Acetate (1 Methoxy-4-vinyloxybenzene); Submitted by: T. Hirabayashi, S. Sakaguchi, Y. Ishii. Checked by: P. W. Davies, A. Fürstner, Org. Synth. 2005, 82, 55.